|
The calculator may continue to prompt you with a message " Number of atoms per molecule has to be a positive integer." This is because this calculator uses atomic masses as listed in the IUPAC atomic weight data, and the values you're using may be slightly different. We recommend you utilize the Atoms per molecule field to calculate the molecular weight, but you're free to manually enter any value in the molecular weight field. To balance a chemical equation, enter an equation of a chemical reaction and press the Balance button. This calculator can support up to five elements per reactant or product. Since there is an equal number of each element in the reactants and products of 4FeS2 + 11O2 2Fe2O3 + 8SO2, the equation is balanced. For example, for H 2 O \text O as its unit. To evaluate the molecular weight, we need to know each element in the compound along with the number of atoms per molecule. Then enter the coefficients, the number of moles, and the mass in the corresponding fields. In this calculator, first set Select calculation type to Calculate molar ratio, moles, and mass. It is possible to calculate the mass of each reactant and product required if you know the molar ratio, number of moles required, and molecular weight of each reactant and product. You will get the resultant molar ratio at the very bottom as a table. In this calculator, first set Select calculation type to Calculate molar ratio and moles and carefully enter the coefficients or the number of moles in the corresponding fields. Since there is an equal number of each element in the reactants and products of H2SO4 H2O + SO3, the equation is balanced. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. Conversely, if you know the number of moles needed, you can compute the molar ratio. Step 4: Substitute Coefficients and Verify Result. 
Since there is an equal number of each element in the reactants and products of 2H2 + O2 2H2O, the equation is balanced. Once you have the molar ratio, you can determine the number of moles of each reactant and product required to complete the reaction. Step 4: Substitute Coefficients and Verify Result. Since there is an equal number of each element in the reactants and products of 3NO2 + H2O 2HNO3 + NO, the equation is balanced. Then carefully enter the coefficients of each reactant and product in the corresponding Coefficient field. Calcium Hydroxide + Phosphoric Acid Tricalcium Phosphate + Water. In addition, the amount of unreacted reagents and the limiting reactant are determined. It also calculates the amounts of reactants and reaction products in both moles and grams. In this molar ratio calculator, first set Select calculation type to Calculate molar ratio. This online Chemical Reaction Calculator checks whether a given chemical equation is balanced and finds the appropriate stoichiometric coefficients. 
Keep in mind that in all these cases, the resultant molar ratio is displayed as a table at the very bottom of the calculator.Ĭalculating the molar ratio from a balanced reaction is the most straightforward method. Question: For the following balanced chemical equation, calculate how many moles of products would be produced if 0. C The actual yield was only 15.7 g of procaine, so the percent yield (via Equation 4.3.5) is. 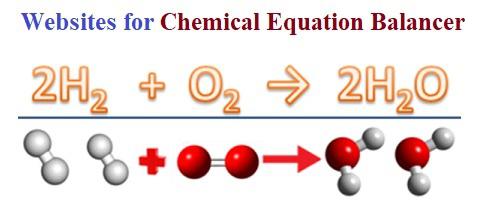
Find more Chemistry widgets in WolframAlpha. Get the free 'Chemical Reaction Calculator' widget for your website, blog, Wordpress, Blogger, or iGoogle. 
theoretical yield of procaine 0.0729mol × 236.31g 1mol 17.2g. Calculator designed to balance chemical equations with results of: the balanced equation, word equation, and how it happened. Additionally, there are three types of calculations to choose from, explained below. To calculate the corresponding mass of procaine, we use its structural formula (C13H20N2O2) to calculate its molar mass, which is 236.31 g/mol. When a chemical equation is balanced it means that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides.This online molar ratio calculator can handle up to five reactants and five products. \): Regardless of the absolute number of molecules involved, the ratios between numbers of molecules of each species that react (the reactants) and molecules of each species that form (the products) are the same and are given in the chemical equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
